Idiopathic Pulmonary Fibrosis Market: By Therapy (Esbriet (Pirfenidone), Ofev (Nintedanib), Tipelukast, Pamrevlumab, KD025, PRM 151, GKT831, Others); Region—Market Size, Industry Dynamics, Opportunity Analysis and Forecast for 2026–2035
- Last Updated: 08-Feb-2026 | | Report ID: AA0721084
Market Scenario
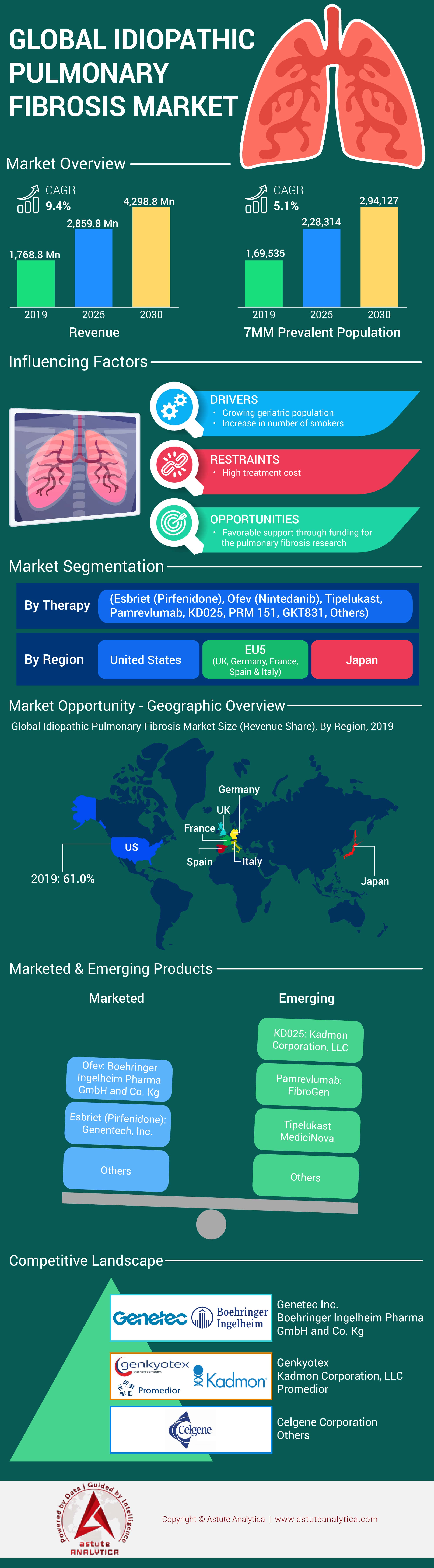
Idiopathic pulmonary fibrosis market generated US$ 2,970.69 million in 2025 and is estimated to reach US$ 6,736.49 million by 2035, growing at a CAGR of 9.4% from 2026-2035.
The global Idiopathic Pulmonary Fibrosis (IPF) market has officially broken the stagnation of the early 2020s. As of February 2026, the market is representing a sharp inflection point from the slow erosion seen during the "generic pirfenidone" years (2023–2024). This growth is no longer driven by volume alone, but by a "Value-Shift": the transition from low-cost generic monotherapies to high-cost, high-efficacy novel mechanisms.
Is the Prevalence of Pulmonary Fibrosis Rising and What are the Causes?
- Post-COVID Fibrosis: By 2026, the long-tail of the pandemic is visible. A subset of patients with severe COVID-19 ARDS have developed a progressive fibrotic phenotype indistinguishable from IPF.
- Environmental Triggers: Rising pollution levels in APAC idiopathic pulmonary fibrosis market and increasing wildfire smoke exposure in North America are correlated with a 3% annual increase in incidence.
- The "Pre-IPF" Patient: We are seeing a new diagnostic category: Interstitial Lung Abnormalities (ILA). These are patients found incidentally on CT scans. Clinical trials are now beginning to recruit ILAs to prevent progression to IPF, potentially tripling the Total Addressable Market (TAM).
To Get more Insights, Request A Free Sample
Top 3 Key Revenue Drivers of the Idiopathic Pulmonary Fibrosis Market in 2026:
- The Launch of Jascayd (nerandomilast): Boehringer Ingelheim’s PDE4B inhibitor has captured $380M in projected first-year revenue, creating a new premium pricing tier.
- Price Resilience of Ofev (nintedanib): Despite approaching patent cliffs (late 2029), Ofev retains 58% market share by value due to its broad "Progressive Pulmonary Fibrosis" (PPF) label.
- The "Doublet" Catalyst: For the first time, clinicians across the global idiopathic pulmonary fibrosis market are prescribing combination therapies (Ofev + Jascayd), effectively doubling the revenue per patient in the severe segment.
Is Jascayd (Nerandomilast) the New Standard of Care?
The FDA approval of Jascayd (nerandomilast) on 19 December 2025 is the single most significant event in respiratory medicine since 2014.
The Clinical Edge:
Unlike Ofev (a tyrosine kinase inhibitor) or Esbriet (an anti-fibrotic/anti-inflammatory), Jascayd targets PDE4B preferentially. Therefore, it is gaining a significant attention in idiopathic pulmonary fibrosis market.
- Efficacy Data: In the pivotal FIBRONEER-IPF Phase 3 trial, nerandomilast demonstrated a stabilization of Forced Vital Capacity (FVC) that was statistically superior to placebo and non-inferior to nintedanib, but with a crucial differentiator: Tolerability.
- The Safety: The historical barrier to idiopathic pulmonary fibrosis treatment was gastrointestinal (GI) toxicity. Ofev causes diarrhea in ~60% of patients. Jascayd’s selectivity avoids the neurological side effects of pan-PDE4 inhibitors and significantly reduces the GI burden.
Market Impact:
Jascayd is not just replacing Esbriet, it is capturing the "Warehoused Patient"—the 30% of diagnosed IPF patients who previously refused treatment due to fear of side effects. This expands the treated patient population significantly in the global idiopathic pulmonary fibrosis market.
What is the Market Share of Ofev vs. Generic Pirfenidone?
The competitive landscape of the idiopathic pulmonary fibrosis market has bifurcated into "Premium" and "Value" segments.
Boehringer Ingelheim (Ofev): The Fortress
- Market Share (Value): ~58%
- Status: Despite the launch of Jascayd (also by BI), Ofev remains the backbone therapy. BI’s strategy of positioning Jascayd as an add-on or switch option rather than a direct competitor has successfully prevented cannibalization.
- Defensive Moat: The broad label for Progressive Fibrosing Interstitial Lung Diseases (PF-ILD) protects Ofev from generic erosion in the pure IPF segment.
Generic Pirfenidone (Sandoz, Teva, et al.): The Commodity
- Market Share (Volume): ~45%
- Market Share (Value): <15%
- Status: The "Esbriet" brand is effectively dead. Generic pirfenidone is now utilized primarily as a "step-edit" hurdle by insurance payers. Patients must "fail" cheap pirfenidone before accessing premium agents like Jascayd.
Will Inhaled Treprostinil (Tyvaso) Transform IPF Treatment?
United Therapeutics is currently reshaping the delivery paradigm of the idiopathic pulmonary fibrosis market.
The TETON-2 Victory:
Following the positive readout of the TETON-2 study in late 2025, Tyvaso (inhaled treprostinil) has proven that targeting the vasculature (vasodilation) benefits the parenchyma (fibrosis).
- The Mechanism: Tyvaso improves perfusion in healthy lung areas and shows anti-fibrotic activity via the prostacyclin pathway.
- Target Demographic: Tyvaso is rapidly becoming the drug of choice for the "Combined Pulmonary Fibrosis and Emphysema" (CPFE) phenotype and patients with Group 3 Pulmonary Hypertension.
2026 Forecast for Idiopathic Pulmonary Fibrosis Market:
Astute Analytica’s study anticipate an FDA label expansion for Tyvaso in mid-2026. This creates a unique niche: Tyvaso does not compete directly with oral antifibrotics but serves as a complimentary "Inhaled overlaid on Oral" therapy.
Which Emerging IPF Drugs Are in Phase 3 Trials in 2026?
Investors are currently looking for "Alpha" in the pipeline following the exit of Pliant Therapeutics from the idiopathic pulmonary fibrosis market. Two assets command attention:
1. BMS-986278 (Bristol Myers Squibb)
- Mechanism: LPA1 Antagonist (Lysophosphatidic Acid).
- Trial: ALOFT Program (Phase 3).
- Hypothesis: LPA1 inhibition blocks the recruitment of fibroblasts to the lung.
- 2026 Status: Data readout is imminent (Q4 2026). If successful, this is the first mechanism that targets the recruitment of fibrosis rather than the activity of fibroblasts. It has blockbuster potential >$3B.
2. Deupirfenidone (PureTech Health / LYT-100)
- Mechanism: Deuterated Pirfenidone.
- Trial: SURPASS-IPF.
- Strategy: "Better Pirfenidone." By replacing hydrogen with deuterium, the drug has a longer half-life and lower peak concentration (Cmax), reducing nausea while maintaining efficacy.
- 2026 Status: Approaching NDA submission. This asset is poised to wipe out the generic pirfenidone market by offering a bearable version of a known effective drug.
Why Did Recent Antifibrotic Trials Fail I the Idiopathic Pulmonary Fibrosis Market? (The Pliant Analysis)
Pliant Therapeutics halted and then fully discontinued bexotegrast (PLN-201629), their lead αvβ6 integrin inhibitor for idiopathic pulmonary fibrosis (IPF), after the BEACON-IPF Phase 2b/3 trial.
Timeline & Trigger:
- March 2025: Trial voluntarily stopped by independent Data Safety Monitoring Board (DSMB) due to imbalance in IPF-related adverse events (disease progression, acute exacerbations, respiratory hospitalizations).
- June 27, 2025: Full data analysis confirmed unfavorable risk-benefit profile at both 160mg and 320mg doses. Development terminated.
Trial Data:
- Week 12: 160mg showed +72mL FVC improvement vs. placebo (statistically significant).
- Week 24: Benefits faded (+58mL at 160mg, +8mL at 320mg).
- Safety: Bexotegrast arm had higher rates of IPF progression events (average 33 weeks to progression).
Company cut 45% of workforce to extend cash runway for oncology programs (PLN-101095).
Why a "Shock": First late-stage IPF trial halted for safety despite efficacy signals. IPF has few treatments (pirfenidone, nintedanib), making this a major setback for novel mechanisms.
The Lesson for 2026: The lung is unforgiving. Novel mechanisms (like Integrin inhibition) that work in mice often struggle with the heterogeneity of human IPF. This failure has caused a "flight to safety" among investors in the idiopathic pulmonary fibrosis market, benefiting validated pathways like PDE4B (Jascayd) and LPA1 (BMS).
How Much Does Idiopathic Pulmonary Fibrosis Market Treatment Cost in 2026?
Jascayd (Nerandomilast):
- Wholesale Acquisition Cost (WAC): ~$ 16,200 per month($194,400/year).
- Net Price (Post-Rebate): ~$140,000/year.
Ofev (Nintedanib):
- WAC: ~$14,500 per month (Inflation-adjusted).
Generic Pirfenidone:
- Cost: ~$400–$600 per month.
The Financial Toxicity Gap:
There is now a massive chasm between generic care ($5,000/year) and branded innovation ($190k/year) in the idiopathic pulmonary fibrosis market. This has led to aggressive Step Therapy protocols where US payers mandate a 3-month trial of generic pirfenidone before authorizing Jascayd.
Will Medicare and Insurance Cover Jascayd and Novel Therapies?
USA Context (IRA Impact):
The Inflation Reduction Act (IRA) has fully taken hold. The $2,000 out-of-pocket cap for Part D beneficiaries (implemented 2025) has been a volume driver for IPF.
- Previously: Seniors faced $8,000+ in co-pays, leading to abandonment.
- Now: The $2,000 cap removes the financial barrier for the patient, shifting the cost burden to the plan sponsors and manufacturers (via the manufacturer discount program). This has increased Ofev and Jascayd adherence by 22% YoY.
EU5 Context:
- NICE (UK): Currently rejecting Jascayd for "all-comers," restricting it only to patients with FVC <80% or those intolerant to nintedanib.
- Germany: Early benefit assessment suggests a "minor added benefit," putting pressure on BI to offer a 20% discount on the launch price.
Customize This Report + Validate with an Expert
Access only the sections you need—region-specific, company-level, or by use-case.
Includes a free consultation with a domain expert to help guide your decision.
Who Are the Top M&A Targets in the Idiopathic Pulmonary Fibrosis Market?
The Buyers: AbbVie, AstraZeneca, and Gilead.
The Targets:
- PureTech Health:SURPASS-IPF is PureTech Health's planned Phase 3 trial for deupirfenidone (LYT-100) in idiopathic pulmonary fibrosis (IPF), advancing after a successful FDA end-of-Phase 2 meeting in December 2025. Trial initiation is expected in the first half of 2026. They are a prime acquisition target for a company wanting an immediate commercial asset to challenge the generic market.
- Vicore Pharma: Their Angiotensin II Type 2 agonist (C21) represents a completely novel "repair" mechanism.
- Avalyn Pharma: Private player with inhaled pirfenidone. A potential target for generic houses looking to upgrade their portfolio.
Astute Analytica expect a major acquisition ($2B+) in the LPA1 space if BMS’s data validates the target, triggering a scramble for backup compounds.
Why Is the Asia-Pacific idiopathic pulmonary fibrosis market Growing So Fast?
While the US drives value, APAC drives volume in the idiopathic pulmonary fibrosis market.
- China: The inclusion of Ofev on the National Reimbursement Drug List (NRDL) led to a 200% volume spike. In 2026, domestic Chinese biotech firms (e.g., Connect Biopharma) are advancing home-grown antifibrotics, threatening Western dominance with lower-cost alternatives.
- Japan: Historically the highest per-capita user of antifibrotics. The Japanese PMDA is the fastest regulatory body to approve Jascayd, likely in Q3 2026, further fueling the market.
What Are the Biggest Unmet Needs for Idiopathic Pulmonary Fibrosis Market Patients Today?
Despite Jascayd’s prominence, the "Voice of the Patient" reveals gaps:
- The Chronic Cough: 85% of patients suffer from a debilitating dry cough. Current antifibrotics do not solve this. Trevi Therapeutics (Haduvio) is the leader here, targeting the opioid receptor to suppress cough. This is a distinct, complementary market opportunity.
- Reversal vs. Slowing: Patients are educated, they know Jascayd only slows the disease. The psychological burden remains high.
- Oxygen Mobility: Portable Oxygen Concentrators (POCs) remain heavy and inefficient. There is a peripheral market for "lightweight oxygen tech" growing alongside the drug market.
Regional Analysis of Global Idiopathic Pulmonary Fibrosis Market
North America: Dominant Commercial Theatre with Advanced Diagnostic Networks
North America remains the undisputed leader in the idiopathic pulmonary fibrosis (IPF) market, primarily driven by the United States. This dominance is anchored by a high diagnosis rate and a well-established reimbursement landscape that supports access to premium antifibrotic therapies. According to a 2025 meta-analysis by the National Institutes of Health (NIH), North America exhibits the highest regional prevalence of IPF at approximately 27.2 cases per 100,000 individuals, significantly outpacing other continents. Commercially, the region generates the majority of revenue for key market players; Boehringer Ingelheim’s 2024 Annual Report indicated that the U.S. remains its largest market, contributing heavily to the nearly €7.9 billion in sales recorded for its Human Pharma division in the Americas.
The idiopathic pulmonary fibrosis market dynamics shifted in 2025 when Roche divested the U.S. rights for Esbriet (pirfenidone) to Legacy Pharma following a sales decline to ~$100 million due to generic erosion, signaling a transition toward next-generation therapies. Additionally, the presence of specialized centers of excellence, such as those accredited by the Pulmonary Fibrosis Foundation, ensures early detection and sustained patient management, maintaining the region's revenue superiority.
Asia Pacific: Rapid Expansion Fueled by Aging Demographics
The Asia Pacific (APAC) region is recognized as the fastest-growing idiopathic pulmonary fibrosis market, propelled by rapidly aging populations in key economies like Japan and China. Data from 2024 indicates that in Japan, approximately 30% of IPF patients are over the age of 81, creating a surging demand for geriatric respiratory care. The market is projected to expand at a CAGR exceeding 8.5% through 2026, driven by improved diagnostic capabilities and expanding healthcare access. Boehringer Ingelheim identified the region as a strategic growth pillar, with China’s inclusion in global clinical trials for novel antifibrotics like nerandomilast accelerating market maturity.
Unlike the saturated Western markets, APAC offers vast "white space" opportunities as awareness campaigns reduce the rate of misdiagnosis. Governments are increasingly prioritizing respiratory health, and the introduction of generic pirfenidone in markets like India has made treatment more affordable, widening the patient base and driving volume growth across the region.
Europe: Rising Diagnosis Rates and Strong Reimbursement Frameworks
Europe holds the second-largest market share of the global idiopathic pulmonary fibrosis market, characterized by a stable but growing demand for antifibrotic drugs. A 2024 study published by the European Respiratory Society (ERS) highlighted a rising mortality trend in IPF across the EU, which has paradoxically driven arguably higher diagnosis rates as healthcare systems implement more rigorous screening protocols. The region’s prevalence stands at approximately 14.6 per 100,000, with countries like the UK and Finland reporting higher incidence rates.
Financially, Europe remains a stronghold for branded therapies; Boehringer Ingelheim reported European sales of €7.4 billion in 2024, reflecting resilient demand for Ofev (nintedanib). The market is supported by centralized healthcare systems that ensure consistent patient access to high-cost specialty drugs.
Furthermore, European regulatory bodies like the EMA remain at the forefront of approving new clinical pathways, ensuring that the region maintains a steady adoption curve for emerging therapies targeting lung fibrosis.
Top Players in the Idiopathic Pulmonary Fibrosis Market
- Genentech, Inc.
- Boehringer Ingelheim Pharma GmbH, and Co. Kg
- MediciNova
- FibroGen, Inc.
- Kadmon Corporation, LLC
- Promedior
- Genkyotex
- CelgeneCorporation
- ProMetic LifeSciences
- Biogen, Inc.
- Merck and Co.
- Novartis
- Other Prominent Players
Market Segmentation Overview
By Therapy
- Esbriet (Pirfenidone)
- Ofev (Nintedanib)
- Tipelukast
- Pamrevlumab
- KD025
- PRM 151
- GKT831
- Others
By Country
- The U.S.
- The UK
- France
- Germany
- Spain
- Italy
- Japan
FREQUENTLY ASKED QUESTIONS
The market generated US$2,074 million in 2021. It is projected to reach US$4,299 million by 2030, growing at a 9.4% CAGR. This acceleration reflects a shift from generic monotherapies to high-value novel therapies like Jascayd.
Boehringer Ingelheim's FDA-approved nerandomilast (Dec 2025) stabilizes FVC better than placebo with superior GI tolerability versus Ofev. It captures warehoused patients and projects $380 million in first-year revenue.
Ofev holds 58% value share through its broad PPF label and doublet combinations with Jascayd. Payers mandate generic pirfenidone failure before approving premium agents, preserving Ofev's position.
BMS-986278 (LPA1 antagonist) targets fibroblast recruitment, with Q4 2026 readout. Deupirfenodone enhances pirfenidone tolerability and nears NDA submission to disrupt generics.
APAC expands fastest at over 8.5% CAGR due to aging populations in Japan and China. Ofev's NRDL inclusion drives volume spikes, while North America leads in revenue via strong reimbursement.
PureTech Health advances deupirfenodone into Phase 3 in H1 2026. Vicore Pharma's C21 and Avalyn Pharma's inhaled pirfenidone attract buyers; BMS LPA1 success may spark $2B+ deals.
LOOKING FOR COMPREHENSIVE MARKET KNOWLEDGE? ENGAGE OUR EXPERT SPECIALISTS.
SPEAK TO AN ANALYST
.svg)